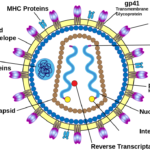
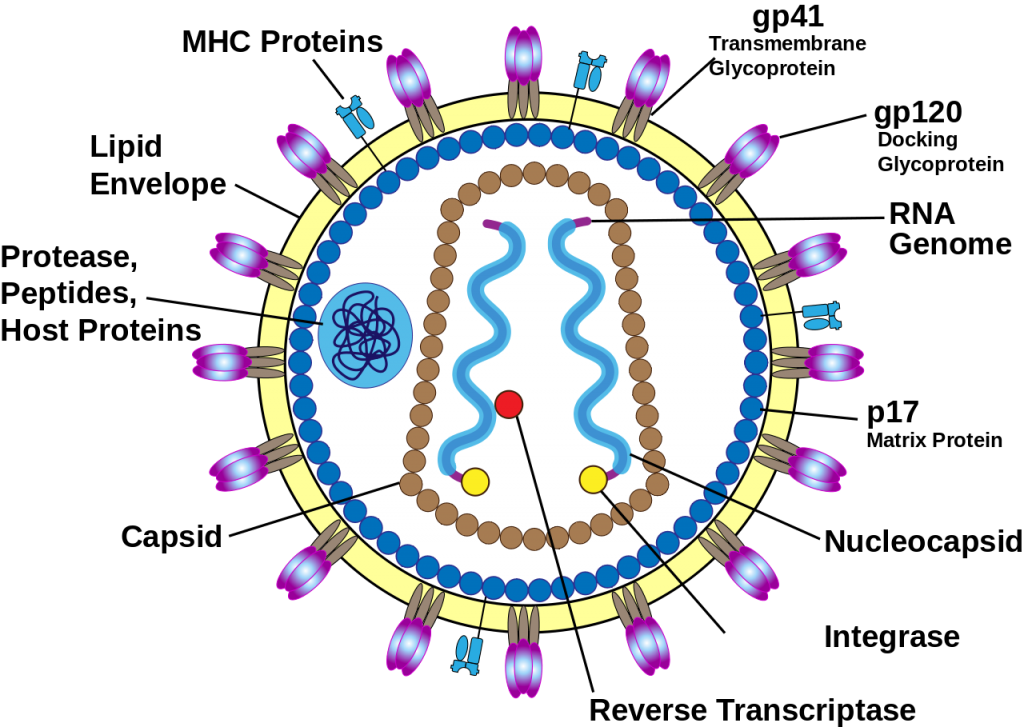
The HIV virus has a spherical structure about 100 to 120 nm in diameter, featuring a lipid envelope, protein matrix, conical capsid, and internal RNA genome with enzymes.
Outer Envelope
The envelope derives from the host cell membrane and contains glycoprotein spikes: gp120 (surface unit for host cell attachment) and gp41 (transmembrane unit for fusion). These env gene products enable entry into CD4 + T cells.
This colourful diagram shows the envelope with protruding glycoprotein spikes surrounding the inner core.
Matrix Layer
Beneath the envelope lies the matrix, made of p17 (MA) protein, which stabilizes the structure and links envelope proteins to the core.
It supports virion integrity during assembly.
Capsid Core
The cone-shaped capsid, formed by ~1500 p24 (CA) proteins in hexameric / pentameric rings, protects the genome.
It houses two copies of positive-sense single-stranded RNA (~9 to 10 kb), plus the enzymes reverse transcriptase (RT), integrase (IN), and protease (PR).
This illustration contrasts immature (spherical) and mature (conical capsid) HIV forms, highlighting p14 and internal components.
Genome and Enzymes
The RNA dimer encodes gag (structural: p24, p17, p7 nucleocapsid/NC, and p6), pol (enzymes), and accessory genes (vif, vpr, tat, rev, nef, and vpu for HIV-1).
NC protein binds RNA; enzymes enable replication via reverse transcription.
Accessory Proteins
Proteins like Vif, Vpr, and Nef enhance infectivity, evade immunity, or regulate expression.
HIV-1 and HIV-2 vary slightly in accessory genes.
How Does HIV Attach to and Enter Host Cell
HIV attaches to host cells primarily through its envelope glycoprotein gp120 binding to the CD4 receptor on target cells like T lymphocytes, followed by interaction with a co-receptor (CCR5 or CXCR4), triggering membrane fusion via gp41.
Attachment Process
GP20 on the viral envelope first binds CD4, inducing a conformational change that exposes the co-receptor binding site.
This high-affinity interaction occurs on CD4+ T cells, macrophages, and dendritic cells, with initial low-affinity contacts via host proteins like heparin sulfate aiding approach.
Co-receptor Engagement
After CD4 binding, gp120 engages CCR5 (for R5-tropic HIV, early infection) or CXCR4 (for X4-tropic, later stages), further reshaping gp120 / gp41.
This primes the fusion machinery, determining viral tropism.
This schematic depicts gp120 binding CD4, then a coreceptor, leading to gp41-mediated fusion and six-helix bundle formation.
Fusion and Entry
GP41’s hydrophobic fusion peptide inserts into the host membrane, pulling viral and cell membranes together to form a six-helix bundle that drives fusion.
The capsid then releases into the cytoplasm in a pH-independent process, mainly direct fusion at the plasma membrane (though endocytosis occurs in some cells).
What Happens After HIV Enters the Host Cell
After entering the host cell cytoplasm via membrane fusion, HIV’s conical capsid releases its contents, initiating reverse transcription of the viral RNA genome into DNA.
Reverse Transcription
The enzyme reverse transcriptase (RT), carried within the capsid, converts the single-stranded RNA genome into double-stranded DNA using host nucleotides; this process occurs partly in the cytoplasm and involves an RNA-DNA hybrid intermediate.
The resulting DNA is complexed with viral proteins in a pre-integration complex.
Nuclear Import
The pre-integration complex, containing viral DNA, integrase (IN), and matrix proteins, traffics to the nucleus via microtubules and host factors like CPSF6 and transportin-1.
It crosses the nuclear pore in a manner dependent on cell division status for some targets.
Integration
The integrase enzyme cuts the host DNA and inserts the viral DNA into the host genome, forming a provirus that can remain latent.
This integrated DNA uses host machinery for transcription into viral mRNA and genomic RNA.
What is Reverse Transcription in HIV Life Cycle
Reverse transcription is a hallmark step in the HIV life cycle where the virus converts its single-stranded RNA genome into double-stranded DNA inside the host cell cytoplasm.
After viral entry, reverse transcriptase (RT), a virally encoded enzyme with DNA polymerase and RNase H activities, uses a host tRNA (typically tRNAys3) primer annealed to the primer binding site (PBS) near the 5’ end of the viral RNA to initiate minus strand DNA synthesis.
RNase H degrades the RNA template as synthesis proceeds, enabling strand transfers: first after strong-stop DNA formation (copying the R-U5-PBS region), then after extending to complete the full-length minus strand DNA using polypurine tract (PPT) fragments.
Key Steps
Initiation
tRNA primer binds PBS; RT synthesizes short minus strand DNA to 5’ R repeat.
First Strand Transfer
Strong stop DNA jumps to the 3’ R repeat.
Elongation and Second Transfer
The minus strand completes; the plus strand initiates from PPT, followed by the second transfer.
This 3D model highlights HIV-1 RT’s p66/p51 heterodimer structure with polymerase (palm, fingers, thumb) and RNase H domains essential for the process.
Final Viral DNA Formation
This result is blunt-ended, linear double-stranded viral DNA (~~9 to 10 kb) within a pre-integration complex ready for nuclear import and integration.
This error-prone process enables high mutation rates and drug resistance.