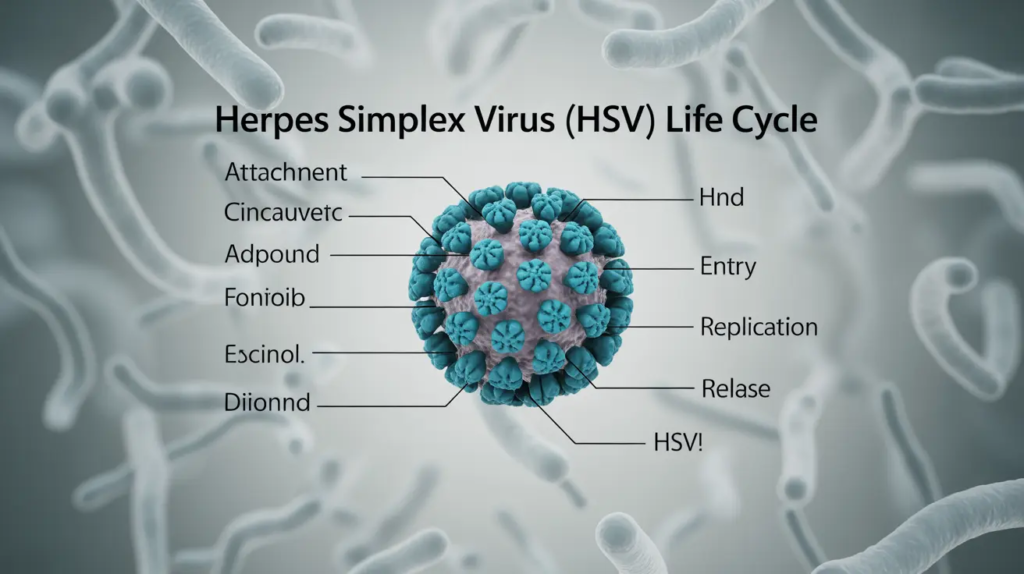
The life cycle of herpes simplex virus (HSV) is generally divided into four main phases: attachment and entry, lytic (productive) replication, establishment of latency, and reactivation.
Attachment and Entry
HSV first binds to receptors on the surface of host epithelial cells, e.g., oral or genital mucosa, using glycoproteins such as gB, gC, and gD; it then fuses with the cell membrane and delivers its nucleocapsid into the cytoplasm. The capsid is transported along microtubules to the nucleus, where the viral DNA is released into the nuclear compartment for transcription and replication.
Lytic (Productive) Replication
After entry into the nucleus, HSV follows a “cascade” of gene expression: immediate-early -> early -> late genes.
- Immediate-early proteins, e.g. ICP4 regulates transcription of early genes.
- Early proteins handle DNA synthesis, e.g., DNA polymerase.
- Late proteins form structural components such as capsid and envelope glycoproteins.
The newly assembled nucleocapsid buds through the inner nuclear membrane, acquiring an envelope and tegument, and then moves through the endoplasmic reticulum and Golgi apparatus, where glycoproteins are processed. Mature virions are transported to the cell surface in vesicles and released by exocytosis or cell lysis, often causing host cell death.
Establishment of Latency
Following primary infection in epithelial cells, HSV-1 and HSV-2 enter the nearby sensory nerve endings and undergo retrograde axonal transport to the cell bodies in sensory ganglia (e.g., trigeminal ganglia for HSV-1 and sacral ganglia for HSV-2).
In these neurons, the viral genome persists as a circular episome with minimal or no production of structural proteins, a state known as latency.
Reactivation and Recurrent Disease
Various stressors (sunlight, fever, immunosuppression, emotional stress, etc.) can trigger reactivation of latent HSV. The virus resumes lytic replication in the ganglion, and newly formed virions are transported anterogradely along the axon back to the original dermatome, where they infect epithelial cells and cause recurrent lesions or asymptomatic shedding.
This cycle of latency and intermittent reactivation allows HSV to persist for the lifetime of the host.
How Does HSV-1 Differ From HSV-2 in the Life Cycle?
HSV-1 and HSV-2 differ mainly in their tissue tropism, preferred ganglia for latency, and frequency of reactivation, while their basic life cycle steps (attachment, entry, lytic replication, and latency) are very similar.
Site of Primary Infection and Tropism
HSV-1 primarily infects oral and facial epithelium (cold sores, keratitis), although it can also cause genital herpes.
HSV-2 shows stronger genital tropism and is the main cause of genital herpes, though it sometimes infects oral sites.
Both enter via mucosal surfaces or abraded skin, bind to similar receptors (e.g., nectin-1, HVEM), and use the same general entry and replication machinery.
Ganglia in Which Latency Is Established
HSV-1: After oral/facial infection, it typically establishes latency in the trigeminal ganglia. After ocular infection, it can also reside in trigeminal or other cranial nerve ganglia.
HSV-2: After genital infection, it usually latently infects sacral dorsal root ganglia and can also reach nearby sensory ganglia.
In both types the genome persists as an episome in sensory neurons with minimal gene expression during latency.
Reactivation Patterns and Disease
HSV-1: Reactivations are common but often less frequent and milder in the genital tract; oral reactivations, e.g., cold sores, are usually short-lived and recurrent.
HSV-2: Causes more frequent and clinically significant recurrences in the genital area, especially after primary infection.
HSV-2 is also associated with higher rates of asymptomatic shedding, which enhances its transmission, while HSV-1 shedding outside the oral region is usually less frequent.
Small Molecular and Immunological Nuances
Molecularly, HSV-1 and HSV-2 share similar immediate-early, early, and late gene expression cascades, but promoter differences (e.g., in the VP16 locus) contribute to distinct latency/reactivation behaviors and immune-evasion profiles.
Some studies suggest HSV-1 spread into neurons is more influenced by T-cell responses than HSV-2, which may partly explain why HSV-2 tends to be more virulent in genital infection models.
Thus, the core life cycle steps are almost identical, but HSV-1 and HSV-2 differ in preferred infection sites, sanctuary ganglia, and the clinical patterns of reactivation and shedding.
Morphology of Herpes Simplex Virus
Herpes simplex virus (HSV) has a complex, enveloped, multi-layered morphology typical of herpesviruses, with a double-stranded DNA genome surrounded by a capsid, tegument, and glycoprotein-studded envelope.
Overall Shape and Size
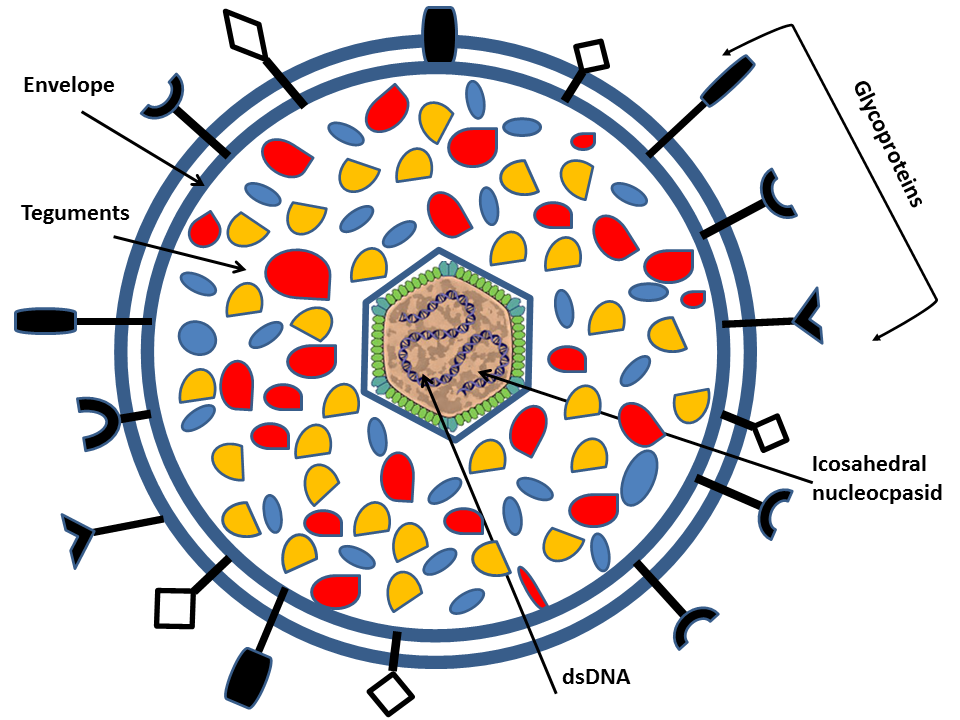
HSV-1 is a large, roughly spherical or pleomorphic enveloped virus about 120–200 nm in diameter when measured by EM, with spikes extending the apparent size up to ~225 nm.
The intact infectious particle is called a virion and shows a clearly defined outer envelope, tegument, and dense nucleocapsid in electron micrographs.
Thus, HSV is a large, enveloped, double-stranded DNA virus with an icosahedral nucleocapsid, an internal tegument layer, and an outer glycoprotein-bearing envelope, giving it the classic “bag-of-spikes” appearance under electron microscopy.